文章
High-resolution, multicolor 3D imaging at the speed of life
An innovative laser fluorescence technique, swept confocally aligned planar excitation (SCAPE) microscopy,
overcomes the limitations of earlier methods and offers broad life sciences utility.
概述
Researchers in diverse fields of life sciences have a common need for 3D fluorescence microscopy tools that are characterized by high speed, high pixel count, and single-cell resolution and can acquire images without causing significant photodamage to the specimen. Despite numerous developments and technical improvements, however, most established techniques still involve tradeoffs that compromise at least one of these parameters.
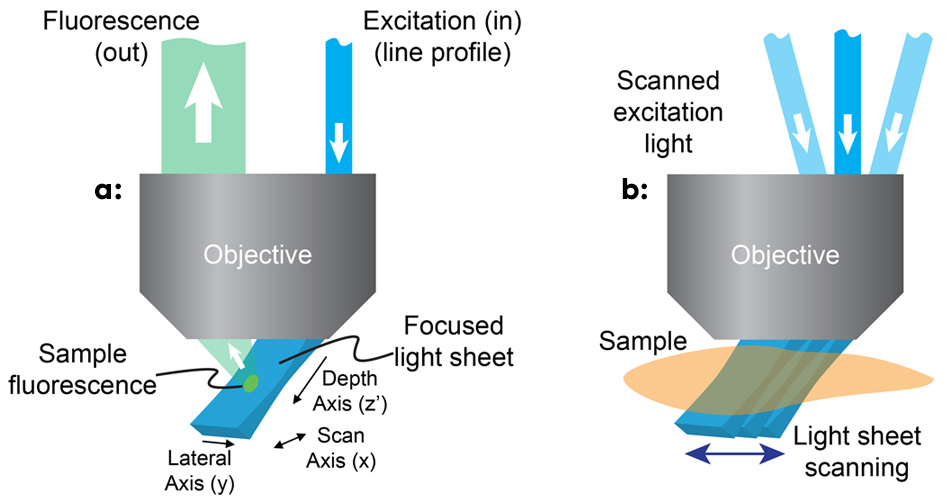
Figure 1: In SCAPE, a light sheet is formed at an oblique angle by off-axis illumination of the primary microscope objective with a line profile beam (a); SCAPE builds a volumetric image by scanning the light sheet while capturing a series of images of the illuminated plane (b).
Progress and tradeoffs
For example, confocal microscopy cannot image a large xyz volume in high resolution at multihertz repetition rates because of the speed limitations of physically scanning a single spot. Moreover, the short dwell time per pixel means that the fastest confocal scans require high laser power, resulting in significant photodamage to live samples.
And while two-photon microscopy dramatically reduces photodamage, such single-point approaches face the same problems resulting from speed/resolution/volume tradeoffs. Recently developed fast acousto-optic modulators (AOMs) now enable faster scanning of small pre-selected volumes, but this approach is of limited use for either large volumes or moving organisms.
Conventional light sheet microscopy enables an entire xy plane to be sampled simultaneously, but it requires lateral sample access (and therefore special preparation) as well as time to build up a cube of 3D data. In addition, synchronization of optics and stage motion make these techniques complex and slow.
Professor Elizabeth Hillman and her colleagues at Columbia University’s Zuckerman Mind Brain Behavior Institute (New York, NY) set out to develop an innovative approach that avoids these limitations while supporting a range of mounted and unmounted sample geometries. Their successful result is swept confocally aligned planar excitation (SCAPE) microscopy, which was first described in a 2015 publication.1 An updated version, SCAPE 2.0, was reported in 2019,2 and Leica Microsystems, recognizing its broad life sciences utility, has now licensed it.
How SCAPE works
希尔曼解释道:“我们认为,真正的高速成像恐怕永远无法通过单点或多点扫描实现。即使我们能达到所需的扫描速度,每个像素的驻留时间也会太短,无法获得信噪比令人满意的图像。因此,我们开始考虑光片显微镜技术。 当时几乎所有系统都需要在样品周围以90°角布置两个物镜。于是问题变成了:我们能否在单物镜配置中结合光片成像的多像素优势?”
研究团队意识到,利用高数值孔径物镜边缘的离轴光路,可以生成一个与显微镜真实xy平面成约45°角的激发光片(见图1)。 为了成像来自该倾斜平面的荧光,他们旋转了物镜的成像平面,以精确对焦相机,其方法类似于倾斜平面显微镜。3 Hillman 及其团队在物镜上游使用一个扫描镜来左右移动光片,该扫描镜还会重新定向返回的荧光,以保持对移动光片的对焦。 随着镜子的移动,通过叠加各平面,显微镜可以快速、反复地生成三维体积图像。
SCAPE 2.0(见图2)的几个细节值得说明。 针对倾斜平面成像(即光片与观察轴成一定角度)的问题,本方案通过使用第二枚物镜将捕获的荧光信号进行中继,从而在中间点形成一个真实的倾斜图像。随后,该图像通过第二枚物镜进行捕获,该物镜以特定角度(约127°)布置,将光片平面平坦地聚焦到相机上。
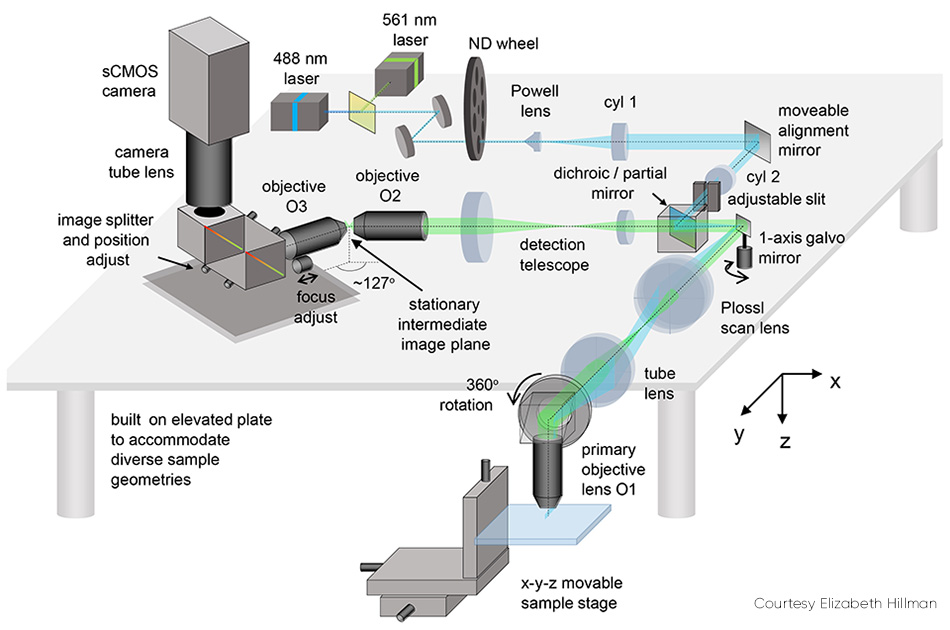
图2:可移动对准镜是SCAPE 2.0的关键组件之一。
相机最终成像的是样品内部的一个倾斜的 y-z 平面,通常呈矩形;由于光线穿透大多数组织的能力有限,该平面在 z 方向(相对于 y 方向)上较窄。 对于此类样本,将相机设置为仅读取较少行数(对应于z方向的深度)非常有用,因为这能实现更快的成像速度——例如,根据所用相机的不同,可以以每秒1000至18,000帧的速度读取200行。
The issue of scan synchronization was first solved by scanning the light sheet using a polygon mirror. The detection path involved the facet adjacent to the one used by the excitation light. Hillman explains, “This polygon was the original inspiration for SCAPE, but we soon realized that it was simpler and just as effective to use a single galvanometer mirror. This change makes the system easier and cheaper to build, lets more light return to the camera, and makes it easier to control the system’s scan patterns.”
With no moving parts other than a galvo mirror, SCAPE’s overall speed is limited only by the camera frame rate and signal-to-noise ratio (SNR). Depending on the specific experiment, the galvo mirror is scanned at between 10 and 100 Hz, corresponding to an unprecedented 10–100 volumes per second (vps). SCAPE uses a conventional sawtooth scan pattern—that is, linear sweep followed by near-instantaneous reset. The amplitude of the galvo’s sweep, and the number of camera frames per sweep, determine the system’s field of view and sampling density in the x direction. Faster cameras can be leveraged to increase volume rates, sampling density, or field of view. While most of the team’s imaging has used standard sCMOS cameras, they reached over 300 vps imaging using an ultrafast CMOS camera with an integrated intensifier.
由于光片是以与图像观察轴 z 成一定角度的方式扫描的,因此每一层深度切片相对于下一层都会产生轻微的偏移。显微镜的计算机通过简单的变换来校正这种“倾斜”,从而生成一个无畸变的 3D 图像体。
数字激光调制
Concurrent monitoring of multiple fluorophores, including functional indicators and fluorescent proteins, enables correlation of dynamic behavior (for example, muscle action) with molecular composition, cellular structure, neural signals, and so forth. SCAPE readily supports such applications by providing multiwavelength excitation—via plug-and-play Coherent OBIS optically pumped semiconductor lasers (OPSLs)—with the option to simultaneously acquire two or more spectrally separated images side by side on the camera.
Hillman cites several innovative advantages of OPSL technology for this work compared to earlier laser types. She notes the wide range of available wavelengths and power levels. “Years ago, we had 488 nm, 532 nm, and 638 nm, and that was about it if you wanted usable power levels. We had no options in the yellow and orange. But today, we can choose laser sources with tens and hundreds of milliwatts at wavelengths that closely match the excitation of almost any commonly available fluorophore.” She explains that most of their SCAPE systems integrate multiple free-space lasers, giving them more flexibility than fiber coupling. “It is super convenient that the lasers are compact and all have identical form factors and the same electronic interface.” To date, Hillman says, they have used as many as five laser wavelengths in some experiments. She also explains that she regularly takes SCAPE to workshops and courses and uses available OBIS lasers with minimal realignment.
Digital imaging is another important feature of OPSLs. Because the OPSLs can be turned on and off at speeds up to 25 kHz, excitation wavelength can be alternated on consecutive frames with precise timing. This is complemented by multiwavelength detection using a lab-built image-splitter composed of dichroic filters and mirrors. This device projects the spectrally separated images with fields of view up to 1280 voxels wide, with no effect on imaging speed compared to single-wavelength operation.
展现动力与续航能力
最近的两项合作研究展示了SCAPE的强大功能及其广泛的应用范围。
对微小生物(包括其全身、大脑和神经系统)进行成像,是神经科学领域的一大趋势。希尔曼及其同事最近发表了一项研究,描述了他们对活体果蝇幼虫中基因编码的钙敏荧光蛋白进行高速三维成像的情况(见图3)。 除了捕捉幼虫在蠕动爬行过程中身体和神经系统的复杂动态变化外,该团队还追踪了体壁神经元在身体变形时的放电情况。
该团队还利用SCAPE技术,研究了活体啮齿类动物皮层中神经元树突的动态放电活动⁵,以及小鼠鼻腔中嗅觉感觉神经元的动态放电活动⁶,并成功对自由活动的秀丽隐杆线虫进行了整体成像。此外,他们还拍摄到了斑马鱼胚胎心脏搏动的高清视频²。
Studies of the embryonic zebrafish heart can provide insights into vertebrate heart development, including the influence of genetic and environmental factors on structure and function. Conventional microscopy requires time gating that inevitably misses details such as irregular arrhythmias given the natural heart rate of 2 to 4 Hz, and it cannot perform full 4D particle tracking for red blood cell (RBC) flow analysis. Hillman’s team partnered with pediatric cardiologist Professor Kimara Targoff, whose lab uses zebrafish to decipher genetic mutations that can cause heart malformations in the embryo. The collaborative effort captured videos of both red blood cells coursing through the beating heart at over 100 vps, and leveraged GCaMP labeling to capture individual waves of calcium activity coursing across the beating heart (see Fig. 4).
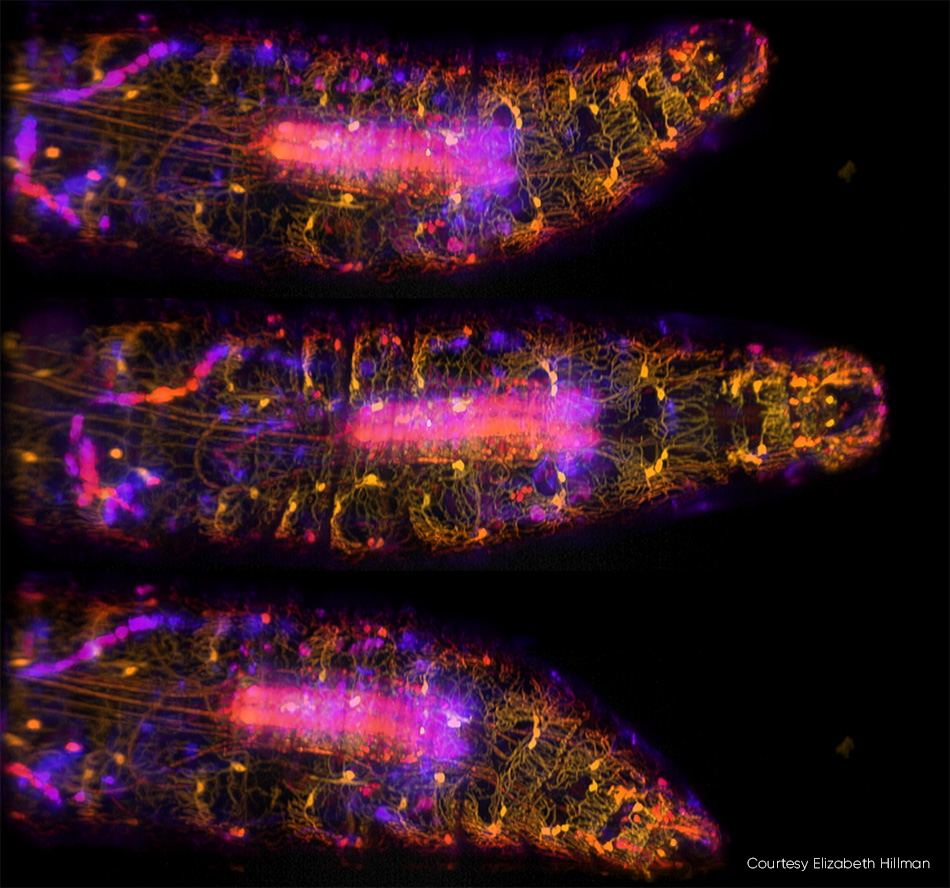
图3:在这三张由SCAPE 2.0以每秒10帧的速度拍摄的果蝇幼虫运动图像[3]中,腹侧本体感觉神经元被标记为GFP,并使用488 nm激发光进行成像。 颜色(从黄色到蓝色)表示来自样本不同深度的信号。详细信息请参见 R. Vaadia 等人的研究 [4];该研究的实时视频序列请访问 http://bit.ly/SCAPE2019。
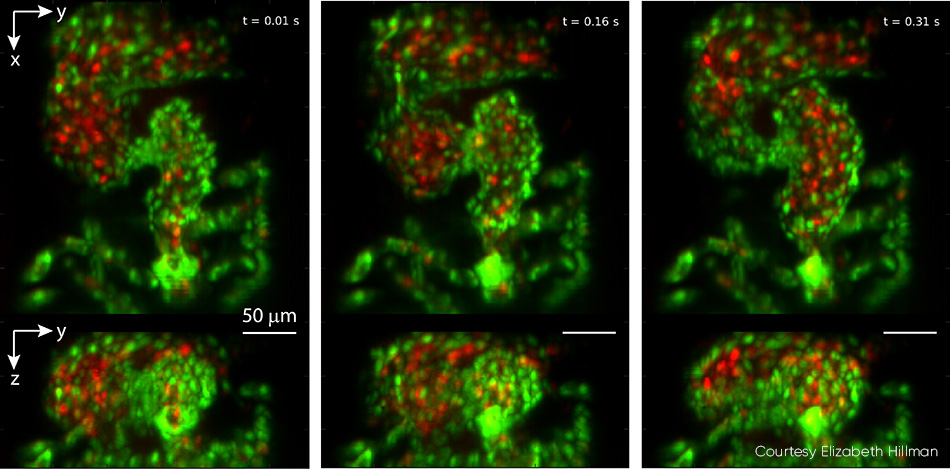
Figure 4: This triptych, drawn from video showing a zebrafish heart beating in real time, was captured at 100 vps. Top panels show z-projection and bottom panels show x-projection. The heart’s ventricle begins compressed, with the outflow valve open, and then fills from the atrium in successive images. Endothelial cells of the heart wall are labeled with EGFP (green) while red blood cells are labelled with DsRed (red). Both fluorophores were excited with 488 nm laser light (0.6 mW at the sample). For full details, including video, see V. Voleti et al. [2]
摘要
Across life sciences, fluorescence microscopy is used as a tool that allows researchers to connect events at the molecular, cellular, organ, and organism levels. The ability to record high-resolution multicolor (3D) images at the speed of life—4D microscopy—is now poised to play a key role in accelerating this research.
参考文献
参考文献
1. M. B. Bouchard et al., Nat. Photonics, 9, 2, 113–119 (2015).
2. V. Voleti et al., Nat. Methods, 16, 10, 1054–1062 (2019).
3. C. Dunsby, Opt. Express, 16, 25, 20306–20316 (2008).
4. R. Vaadia et al., bioRxiv, 467274 (2018).
5. E. M. Hillman et al., Curr. Opin. Neurobiol., 50, 190–200 (2018).
6. L. Xu et al., Science, 368, 6487, eaaz5390 (2020).